What’s going on with the AstraZeneca vaccine?
A number of announcements have recently been made about use of the Oxford-AstraZeneca Covid-19 vaccine, also known as Vaxzevria. This is due to a possible link between the first dose of the vaccine and a specific type of extremely rare blood clot, occurring alongside a low level of platelets (a component of the blood).
Join 73,000 newsletter subscribers who trust us to check the facts
Sign up to get weekly updates on politics, immigration, health and more.
Subscribe to weekly email newsletters from Full Fact for updates on politics, immigration, health and more. Our fact checks are free to read but not to produce, so you will also get occasional emails about fundraising and other ways you can help. You can unsubscribe at any time. For more information about how we use your data see our Privacy Policy.
What’s the risk?
In the period up until 31 March, 20.2 million doses of the AstraZeneca vaccine had been given in the UK, and the Medicines and Healthcare products Regulatory Agency (MHRA) received 79 reports of this specific type of clot. In 19 of these cases, the person died.
That’s equivalent to an overall risk of roughly 1 in 250,000 of getting this type of clot after vaccination. This is a very broad estimate, so for some people the risk may be higher, and for some it may be lower.
To put this into context, we need to compare it with the likelihood of becoming very ill with Covid-19. But there are several factors to consider when it comes to how likely that is, mainly how likely someone is to catch the disease, and how likely they are to have a poor outcome as a result (which is affected mostly by age and health conditions).
The Winton Centre for Risk and Evidence Communication at Cambridge University has calculated the chances of getting this type of blood clot after the vaccine and compared that to the benefits of being vaccinated, which it chose to demonstrate as the number of admissions to intensive care avoided.
If the chances of catching Covid-19 are low, as they were in March 2021, then the immediate potential for harm from the vaccine for those aged 20-29 slightly outweighs the benefits. Vaccinating 100,000 people of that age will avoid about 0.8 ICU admissions due to Covid-19 in a 16 week period.
For the same age group, the Winton Centre estimated that about 1.1 in 100,000 would get the blood clots. But the risks for both are extremely small.
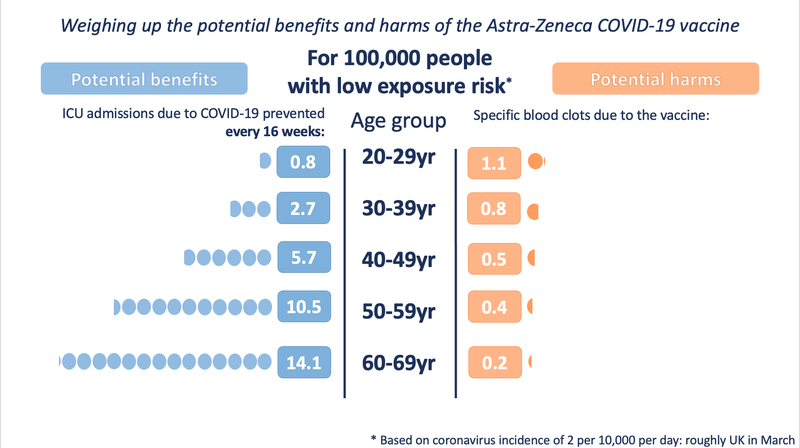
These estimates don’t take into account the other benefits from vaccination, such as avoiding long Covid, or hospitalisation in general.
The benefits of vaccination also rise over time, as the vaccinated person stays protected.
What’s been recommended?
On Wednesday, the MHRA issued a statement, saying that the vaccine was still safe, and that people should go to their vaccine appointments when invited.
It did recommend that those at higher risk of blood clots because of a medical condition should be considered for the AstraZeneca vaccine only if benefits from the protection against Covid-19 infection outweighs these risks. It also says that pregnant women, who are predisposed to thrombosis, should also discuss with their healthcare provider whether the benefits outweigh the risks.
It also said that anyone who had cerebral or other major blood clots occurring with low levels of platelets after their first vaccine dose should not have their second.
Those aged 18-30 will be offered an alternative vaccine
The Joint Committee on Vaccination and Immunisation (JCVI), which advises government health departments on these matters, also recommended that, on balance, it’s “preferable” that adults under 30 (with no underlying health conditions that put them at higher risk of Covid-19) be offered a different Covid-19 vaccine, such as Pfizer or Moderna, if it’s available. It also said such people could make an “informed choice to receive the AstraZeneca COVID-19 vaccine to receive earlier protection.”
On those over 30, the committee said: “JCVI has weighed the relative balance of benefits and risks and advise that the benefits of prompt vaccination with the AstraZeneca COVID-19 vaccine far outweigh the risk of adverse events for individuals 30 years of age and over and those who have underlying health conditions which put them at higher risk of severe COVID-19 disease.”
Is it more likely in women?
Out of the 79 cases, 51 were in women and 28 were in men. Of the 19 deaths, 13 were women and six were men. This could possibly be because many more women have been given the vaccine. In a press briefing on Wednesday, Professor Sir Munir Pirmohamed, Chair of the Committee of Human Medicines, said this and added that many more frontline healthcare workers are women, and from the data so far, the incidence rates for both sexes was the same.
But he also said we don’t yet have enough evidence to say whether these blood clots are more likely to happen in women.
What kind of blood clots are these?
Concern has arisen around cases of a rare type of blood clot that occurs alongside low levels of blood platelets.
Out of the 79 cases, 44 were a type that occurs in the brain called cerebral venous sinus thrombosis, or CVST.
The other 35 cases were blood clots in other major veins, also with low platelets.
What has the EU said?
The European Medicines Agency (EMA) also said on Wednesday that there was a “possible link” between the vaccine and very rare blood clots with low blood platelets, but that overall, the benefits of the vaccine outweighed the risks.
It looked at data from the EU’s drug safety database, which reported 62 cases of cerebral venous sinus thrombosis and 24 cases of splanchnic vein thrombosis (involving one or more abdominal veins) as of 22 March 2021, including in the UK. It said 18 of these cases were fatal.
Overall, the agency recommended that unusual blood clots with low blood platelets should be listed as very rare side effects of this vaccine, but made no specific recommendations as to who should or should not have it.